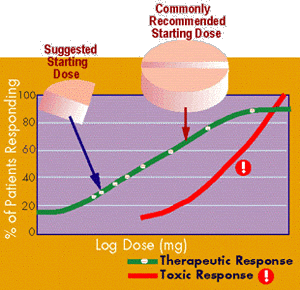

There are numerous examples of recommended introductory doses which are larger than those eventually needed. These larger "recipe" doses increase the chance of adverse effects as well as cost. A classic example, still seen in the 1995 CPS, is the dose recommendation for the antihypertensive drug Hydrodiuril (hydrochlorothiazide) of 50-200 mg/day. We have known for a long time that doses as low as 6.25 mg or 12.5mg/day are often effective and that the dose should rarely exceed 25 mg/day.(1) The 1995 CPS recommended dose for Capoten (captopril) is 25-150 mg TID. We know now that 6.25 mg BID is a good starting dose and doses greater than 25 mg TID are seldom needed. Two years ago, Imitrex (sumatriptan), was marketed in Canada only as a 100 mg tablet. In recognition of the need for lower doses it has just been approved in the USA as a 25 and a 50 mg tablet.
Several factors appear to influence initial drug dosage recommendations.(2) The Federal Health Protection Branch requires that drug companies show that a drug has an "effect", often compared to placebo, before it can be approved. The way to show that a drug has a significant effect in the smallest number of study patients is to use doses of the drug that will work in most study patients. Study patients are highly selected and often include only middle-aged, Caucasian males, who are receiving no other drugs and have no concomitant disease. Once approved, the company must base dosing recommendations on the available study data. Unfortunately, these study patients may have little in common with your patients, particularly the elderly who are more likely to have altered drug handling and responses.
The typical "population" log dose-response curve shown in the Figure provides an example of the cumulative percentage of patients who respond to incremental doses of a drug. It is clear that different patients have similar therapeutic responses to very different drug doses.
Starting at the lower left portion of the efficacy curve, about 25% of patients achieve the desired effect with "low" doses and a very low likelihood of a toxic response. At the top right end of the curve, a point is reached where additional dose increases may not increase the number of people who achieve the desired effect. In other words, there a significant number of patients (10-30%) who are nonresponders and "pushing" the dose will be futile, adding only to adverse effects and cost.
For those patients who do not require an immediate response (hypertension, NIDDM, osteoarthritis, hyperlipidemia), start with a dose that is a 1/4 to 1/2 the recommended starting dose in the CPS. Then titrate the daily dose two to four weeks (if this is a useful interval) to the desired effect. To use this approach, discussion with the patient must take place before starting drug therapy. The patient must be told not to be disappointed if an immediate effect is not seen and that over the next few weeks the dose will be titrated to achieve an effective dose. Tell the patient that this approach reduces the chance of adverse effects and may reduce the cost of the drugs.
A titration approach can also apply to more "acute" conditions such as short-term insomnia, headache, allergies, or the common cold etc. For these "acute" conditions one should start with the lowest available dose and titration should occur over hours to days rather than days to weeks.
This is a real challenge since the tendency is "don't rock the boat." There are ample successful studies of gradual dose reduction and even discontinuation of such agents as antihypertensives(3) and anticonvulsants(4). As long as the dose reduction is small (reduce the dose by 1/4 each time) and at wide enough intervals (to avoid unnecessary office visits), there should be little patient anxiety. If the signs or symptoms reemerge, the diagnosis and the effectiveness of the drug are verified and the dose may again be increased. When patients are symptomatic they can often titrate the dose themselves as long as they are given the appropriate guidelines.
Start with the product monograph recommended dose, hopefully obtaining a response, and later gradually reduce the dose according to the patient's beneficial/toxic effects. The advantage of this approach is that an effect will likely be seen more quickly than with the titrating up approach. However, the titrating down approach is rarely considered since clinicians and patients are often reluctant to decrease the dose if a benefit has been achieved. Even in the face of a drug benefit without signs of toxicity, drug therapy should be slowly tapered to the lowest effective dose. Sometimes this turns out to be no drug at all. This may be the result of an initial incorrect diagnosis, fluctuating disease state, or effective non-drug therapy such as lifestyle changes.
Many tablets and caplets can be broken in 1/2 or ¼ (a community or hospital pharmacist can provide guidelines for specific dosage manipulation). Tablets can be broken either by placing the tablet on a hard surface and pressing down on either side or by getting your patient to purchase a pill cutter (available in most pharmacies). Capsules or hard coated tablets are more difficult to work with. In some cases, even these can be reduced by 1/2 the daily dose by giving the tablet every other day rather than daily (e.g. omeprazole 20 mg every other day). This is only feasible for drugs with a duration of activity in the order of days rather than hours.
For cases where an immediate effect is wanted (acute alcohol withdrawal, severe pain, severe asthma, severe angina, status epilepticus, etc.) or the consequences of underdosing are potentially dangerous (acute bacterial infections), it is worth erring on the side of giving "too much", as long as the drug has an acceptable toxicity profile, rather than using a slow upwards titration.
Due to unpredictable responses in individual patients, the initial prescription for almost all drugs should be roughly 1-2 weeks to prevent unnecessary wastage in the patient who cannot tolerate the drug. The duration of subsequent prescriptions should be based on the need for future follow up. If the patient needs to be reevaluated in 6 weeks the prescription should likely be written for that time period. Once the best dose for a particular patient has been determined and if chronic therapy is required, the duration of the prescription should reflect the requirement for reassessment. This may be 3 months, 6 months or a year depending on the condition being treated and the need for reevaluation.
Please send feedback!
Back to Therapeutics Letter.
![]() Back to the Therapeutics
Initiative Home Page.
Back to the Therapeutics
Initiative Home Page.
©Therapeutics Initiative. Last updated: December 28, 1995.